
It was not until 1974 that Hsps were discovered and interest in this field of biology became widespread ( Tissieres et al., 1974). 
The heat shock response was first discovered in the early 1960s in Drosophila that displayed changes in salivary gland transcriptional activity in response to different incubation temperatures ( Ritossa, 1962). These activities are carried out by molecular chaperones, many of which are heat shock proteins (Hsps). In this review, we will discuss the role of intracellular and extracellular STI1 and the Hsp70/Hsp90 chaperone network in mechanisms underlying protein misfolding in neurodegenerative diseases, with particular focus on AD.īrief Introduction to Chaperones and Co-chaperonesĪ major requirement for cellular growth, function, and survival is the proper folding, maturation, and degradation of proteins. elegans can also increase toxicity in an amyloid model. Interestingly, decreased levels of STI1 in C. Extracellular STI1 can prevent Aβ toxic signaling by (i) interfering with Aβ binding to PrP C and (ii) triggering pro-survival signaling cascades. In addition to its intracellular function, STI1 can be secreted by diverse cell types, including astrocytes and microglia and function as a neurotrophic ligand by triggering signaling via the cellular prion protein (PrP C). Importantly, STI1 has been shown to regulate aggregation of amyloid-like proteins in yeast. Stress-inducible phosphoprotein 1 (STI1), an intracellular co-chaperone, mediates the transfer of clients from Hsp70 to Hsp90. On the other hand, the use of Hsp90 inhibitors in AD mouse models reduces Aβ toxicity, and normalizes synaptic function. For example, knockdown of Hsp90 and its co-chaperones in a Caenorhabditis elegans model of Aβ misfolding leads to increased toxicity. However, the role of Hsp90 in regulating protein misfolding is not yet fully understood. The heat shock protein (Hsp) family, particularly Hsp70 and Hsp90, plays a major part in this process and it is well-known to regulate protein misfolding in a variety of diseases, including tau levels and toxicity in AD. They form an important line of defense against misfolded proteins and are part of the cellular quality control system. Chaperones and co-chaperones regulate protein folding and client maturation, but they also target misfolded or aggregated proteins for refolding or for degradation, mostly by the proteasome. Assembles of beta-amyloid (Aβ) peptide-either soluble (oligomers) or insoluble (plaques) and of tau protein, which form neurofibrillary tangles, are the major hallmarks of AD. The accumulation of misfolded proteins in the human brain is one of the critical features of many neurodegenerative diseases, including Alzheimer's disease (AD). 6Department of Anatomy and Cell Biology, Schulich School of Medicine and Dentistry, University of Western Ontario, London, ON, Canada.5Department of Physiology and Pharmacology, University of Western Ontario, London, ON, Canada. 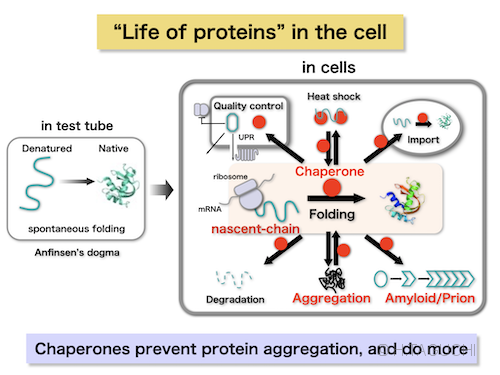
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
